|
|
 |
||||||
A Basic Introduction to Microwave Chemistry© G. Whittaker, 1994 & 2007. This work, or extracts from this work, may be reproduced only with the written permission of the author
The microwave Heating Mechanism
The easiest way to visualise the true mechanism is to picture microwaves for what they are - a high frequency oscillating electric and magnetic fields. Anything that is put into this field, if it may be electrically or magnetically polarised at this oscillation frequency, will be affected. Two principal heating methods exist: Dipolar polarisation, and Conduction mechanisms A third mechanism - interfacial polarisation - occurs, although this is often of limited importance.
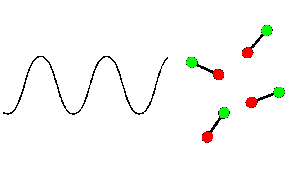
Dipolar PolarisationFor a molecule in a polar liquid such as water (methanol, ethanol, THF, etc.), there are intermolecular forces which give any motion of the molecule some inertia. Under a very high frequency electric field, the polar molecule will attempt to follow the field, but intermolecular inertia stops any significant motion before the field has reversed, and no net motion results. If the frequency of field oscillation is very low, then the molecules will be polarised uniformly, and no random motion results. In the intermediate case, the frequency of the field will be such that the molecules will be almost, but not quite, able to keep in phase with the field polarity. In this case, the random motion resulting as molecules jostle to attempt in vain to follow the field is the heating we observe in the sample.
It is interesting to note that whist the efficiency of microwave absorbance varies markedly with frequency for any liquid, the frequency of a domestic microwave oven (2.45GHz) is NOT selected so that it is at the maximum absorbancy for water (something like 10GHz). If it were you would find that most of the microwave energy was absorbed by the outer layers of your food, whilst the inside stayed unheated and hence uncooked. Note, though, that this is a very simplistic model of microwave heating in hydrogen-bonded systems, which actually involves a complex mechanism that does not simply result from molecular rotations.
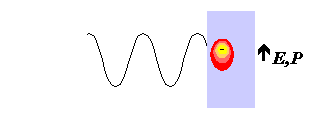
Conduction Mechanisms Where the irradiated sample is an electrical conductor, the charge carriers (electrons, ions, etc.) are moved through the material under the influence of the electric field, E, resulting in a polarisation, P. These induced currents will cause heating in the sample due to any electrical resistance. For a very good conductor, complete polarisation may be achieved in approximately 10-18 seconds, indicating that under the influence of a 2.45GHz microwave, the conducting electrons move precisely in phase with the field.
Thus, if one takes pure water and heats it in a microwave oven, where a variant of the polarisation mechanism dominates, we find that the heating rate is significantly less than when one takes the same volume of water and add salt. In the latter case, both mechanisms occur, and contribute to the heating effect.
Interfacial Polarisation This mechanism is important for systems comprised of conducting inclusions in a second, non-conducting material. An example would be a dispersion of metal particles in, say, sulphur. Sulphur is microwave transparent and metals reflect microwaves yet, curiously, the combination forms an extremely good microwave absorbing material (So good, in fact, that interfacial polarisation effects are reputed to be the basis of 'Stealth' radar absorbant materials). © Gavin Whittaker, 1997. |
||||||